Let’s talk corrosion! There are two types, galvanic and stray current. They are very different and should never be confused.
Galvanic Corrosion
Galvanic corrosion occurs as a result of properties inherent in metals. When immersed in a conductive liquid (an electrolyte, for example saltwater) all metals have a ‘natural’ voltage. If you place two different metals in the same electrolyte and wire them together, the voltage difference will result in a current flow through the connecting wire. Something more complex goes on in the electrolyte but the net effect is the metal with the lower voltage (the anode) is eaten up while the one with the higher voltage (the cathode) is unharmed.
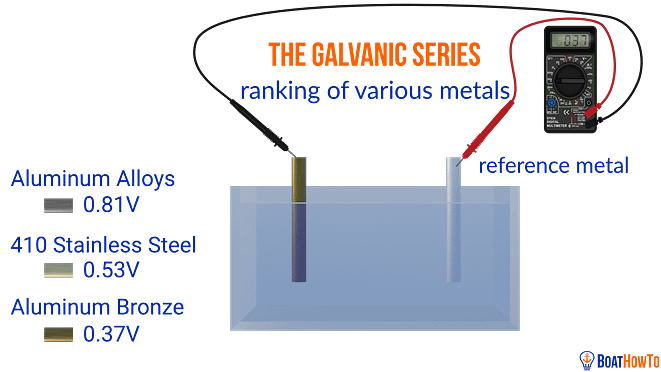
The natural voltage we can measure for various metals with respect to a reference metal
There is a table, the galvanic series table, which ranks metals with respect to their natural voltage, and, as such, their corrosion potential. Note that zinc and aluminum are pretty much at the bottom (most negative and easily corroded end) of this table.
Stray Current Corrosion
Stray current corrosion requires the imposition of an external voltage and current. In this case the natural voltage of the metals involved is irrelevant. Whatever metal is connected to the positive side of the circuit is corroded, and whatever is on the negative side is not, even if it is zinc or aluminum.
Preventing Corrosion
The way we prevent galvanic corrosion is by attaching or wiring a zinc or aluminum sacrificial anode to those underwater metals at risk of corrosion. The sacrificial anode is eaten up, protecting the other metals with a higher natural voltage. The way we prevent stray current corrosion is by ensuring a boat is wired such that no conductor connected to battery positive ever comes into contact with underwater metal. Sacrificial anodes are irrelevant.
The Issue with Thru-Hulls
There was a time when all thru-hulls were manufactured from silicon bronze which is near the top of the galvanic series table and pretty much immune to galvanic corrosion. In general, these thru-hulls needed no additional galvanic protection. But that was many years ago. Today, some still are silicon bronze, especially in the U.S., but a vast number, notably in Europe, are made from some variant of brass.
The core difference between silicon bronze and brass is all forms of brass have some level of zinc in the alloy. Right there, you know there is potentially a problem. Given a connection to a metal without zinc in its alloy, the zinc can be eaten out of the brass leaving a porous and weak fitting likely to fail. Depending on how the brass is alloyed the zinc may be bound up at the molecular level in a manner that reduces this corrosion potential (these alloys are known as ‘dezincification resistant’ – DZR – brass) but the corrosion potential is still there.
Plastic Seacock Options
An alternative to metal seacock assemblies is high-density plastic. Brands like Marelon and Trudesign offer reinforced plastic seacock assemblies that are resistant to corrosion and can withstand significant force without damage or breaking. It's important to choose the right product, as some plastic seacocks are bonded together and cannot be disassembled whereas others can be disassembled. In general, plastic fittings require more space than their bronze or DZR brass equivalents.
To prevent this destruction, we wire all underwater fittings together inside the boat and then connect a chunk of specially alloyed zinc or aluminum into this ‘bonding’ circuit as a sacrificial anode. For this to work, the bonding circuit inside the boat must be near electrically perfect, which means adequately sized conductors, properly installed terminals, and tight, corrosion-free connections to all the thru-hulls being protected.
Sacrificial Anodes
The effectiveness of the immersed sacrificial anode at the other end of the bonding circuit is directly proportional to its surface area. As the anode is consumed, its effectiveness is reduced. The time to replace an anode is when it is half gone. If you wait until it is completely consumed some expensive metal elsewhere on your boat will be corroding!
Historically, we have used zinc as the sacrificial anode in salt water and magnesium in fresh. Today you can buy aluminum anodes which work well in both salt and fresh water. The aluminum lasts longer than zinc and is less harmful to the environment as it dissolves. What you can’t do is mix different anodes in the same bonding system – one will eat up the other.
If you want to know more about various metals and corrosion, check out our Advanced Marine Electrics course. The course includes an entire module dedicated to the topic, where we cover all the important issues.

